African Americans Infected with COVID-19 May Suffer from Sickle Cell Disease, Notes Top Medical Scholar
The effects of the Coronavirus pandemic also known as COVID-19 on the health of racial and other ethnic minority groups such as Hispanics is now gaining the attention of the medical and scientific communities. But more needs to be done in the United States and throughout the world, according to Dr. Uche Sampson, Chief Medical Officer at Micelle BioPharma
 Dr. Sampson noted that the current data suggest a disproportionate burden of illness and death among racial and ethnic minority groups, which includes a number of people who are Hispanic.
Dr. Sampson noted that the current data suggest a disproportionate burden of illness and death among racial and ethnic minority groups, which includes a number of people who are Hispanic.
A recent CDC MMWR report included race and ethnicity data from 580 patients hospitalized with lab-confirmed COVID-19 found that 45% of individuals for whom race or ethnicity data was available were white, compared to 55% of individuals in the surrounding community.
However, 33% of hospitalized patients were black compared to 18% in the community and 8% were Hispanic, compared to 14% in the community.
These data suggest an overrepresentation of blacks among hospitalized patients. Among COVID-19 deaths for which race and ethnicity data were available. New York City pdf iconexternal icon identified death rates among Black/African American persons (92.3 deaths per 100,000 population) and Hispanic/Latino persons (74.3) that were substantially higher than that of white (45.2) or Asian (34.5) persons,” stated the Centers for Disease Control (CDC).
Studies are underway to confirm these data and understand — and potentially reduce — the impact of COVID-19 on the health of racial and ethnic minorities.
 For example, in New York City, African Americans are twice more likely to die than white Americans, according to data released by the city’s Health Department. Other cities are experiencing similar disproportionate mortality rates like Chicago, Detroit, New Orleans and Milwaukee, as are states like Tennessee, Mississippi, and Arkansas.
For example, in New York City, African Americans are twice more likely to die than white Americans, according to data released by the city’s Health Department. Other cities are experiencing similar disproportionate mortality rates like Chicago, Detroit, New Orleans and Milwaukee, as are states like Tennessee, Mississippi, and Arkansas.
During a telephone interview with Conservative Base, Dr. Sampson mentioned he is calling on the federal government to allow the CDC to place a high priority to provide more data regarding the racial and ethnic disparities in COVID-19 outcomes, that many cities are starting to finally recognize.
He is also calling on the federal government, vis-à-vis the CDC, to acknowledge the disparate effect of COVID-19 on the African American community thus enabling serious, targeted health education to minority communities. On both an immediate and long-term basis, efforts need to be redoubled to eliminate the disparities in morbidity and mortality, assure quality health care and eliminate racial disparities in health care and health research.
Dr. Uche K. Sampson, Chief Medical Officer at Micelle BioPharma, Inc wrote a letter to HHS Secretary Alex Azar last month requesting assistance to expedite a Phase 3 trial waiver for the drug Altemia. Altemia has shown to be an incredibly positive treatment for pediatric sickle cell.
Dr. Sampson also stated, “Because this drug has the FDA rare pediatric disease and orphan designations, we hope that the Department will take advantage of the greater flexibility that this allows to ensure that these children have access to it as quickly as possible.”
A well-respected medical practitioner and researcher, Sampson said he is hoping that government agency will reduce the “red tape” that exists with experimental drugs. (See Dr. Sampson’s letter to Secretary Azar of the Health and Human Services Department printed below.)
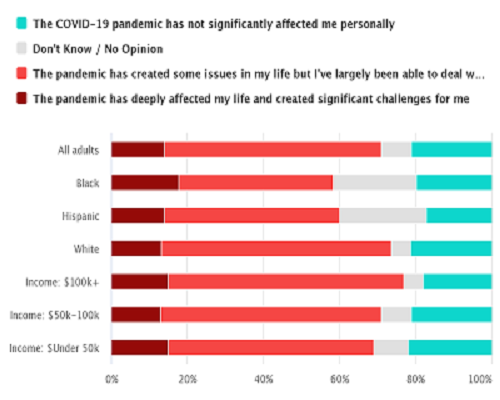
As governmental officials and medical professionals work to mitigate the devastating health consequences of the COVID-19 outbreak, most Americans are personally dealing with secondary effects stemming from the pandemic. Overall, 14 percent of Americans say the pandemic has deeply affected their life and created significant challenges for them. The majority (57 percent) say the pandemic has created some issues in their life but that they are manageable.
Micelle Biopharma is developing a drug called Altemia for sickle cell patients. According to the company’s Dr.Sampson, “The development of Altemia™ is based on the FDA’s findings of safety for Lovaza®, a marketed omega-3 fatty acid product that was approved by the FDA for the treatment of hypertriglyceridemia.”
In other words, Altemia™ is similar to Lovaza®. Both drugs are derived from fish oil, but, Altemia™ dosage is lower.
This drug was already successful from the blinded period of the Phase 2 clinical trial. In response, the FDA supported an Open-Label Extension period of the Phase 2 clinical trial to provide the benefits of the drug to subjects in the placebo arm.
Americans who are susceptible to Sickle Cell Disease need to get this drug as quickly as possible through a Phase 3 trial waiver. Such an action is not unprecedented. In 2015, the FDA approved a Phase 3 waiver for Intellipharmaceutics. This company produced a drug called Rexista™ XR, which is the bioequivalent of OxyContin. If the company was forced to conduct a Phase 3 study, it would have cost almost $20 million dollars. A Phase 3 trial can last as long as three to four years.
CDC Study Corroborates Dr. Uche Sampson’s Statements
A new study of the Coronavirus’ impact on minority communities in the United States – especially African American communities — has corroborated what Dr. Uche Sampson said during his interview with Conservative Base’s editor on May 1.
In the study, the Centers for Disease Control (CDC) authors reported that over 80% of patients hospitalized for the Coronavirus in the state of Georgia in March were African Americans, according to the new CDC study released on Wednesday, April 29, 2020.
The CDC, which is headquartered in Atlanta, Georgia surveyed eight hospitals throughout the Peach State, including seven in the metro Atlanta area and one in southern Georgia.

Among the 305 COVID-19 patients, 247 — more than 80% — were black. Thirty-two patients were white, 10 were Hispanic, and eight were Asian or Pacific Islander.
“The proportion of hospitalized patients who were black was higher than expected based on overall hospital admissions,” the study’s authors wrote.
Black patients weren’t more likely to require a ventilator or die during hospitalization, according to the study. They also weren’t more likely to have diabetes or heart disease, which could affect the severity of COVID-19, though about 74% of patients overall had conditions considered high-risk for severe COVID-19. Diabetes was most common in patients between ages 50 and 64.
“Given the overrepresentation of black patients within this hospitalized cohort, it is important for public health officials to ensure that prevention activities prioritize communities and racial/ethnic groups most affected by COVID-19,” the CDC authors wrote.
About 36% of COVID-19 cases in Georgia are in African American people, according to the state’s public health department, which is a little higher than the state population of 32%.
Similar trends have been documented in other states, according to CBS News.
In Maryland, black people make up 31% of the population and 45% of coronavirus deaths. In Louisiana, black people make up 33% of the population and 56% of coronavirus deaths.
Major cities such as New York, Detroit, and Washington, D.C., have also reported racial disparities in COVID-19 cases, CBS said
The Honorable Alex Azar
Secretary
Department of Health and Human Services
Hubert H. Humphrey Building
200 Independence Avenue, SW, Room 120F
Washington, DC
Dear Mr. Secretary,
Deep-rooted patriotism warrants that able individuals should help confront the tide in the affairs of our nation. Upon us now, is such a tide in the name of the COVID-19 pandemic.
This sentiment is the impetus for seeking your kind consideration and assistance via this medium.
This email bears two attachments. The first is a letter that details our quest while the second provides brief scientific information on the substance that underpins that quest. Collectively, both documents provide concrete rationale for the assistance we seek in effort to contribute our quota towards to success of our nation, particularly in these challenging times.
We look forward to your kind response.
Sincerely,
Uche K. Sampson, MD, MPH, MBA, FACP
Chief Medical Officer
Micelle BioPharma, Inc.
Watch New Video: Opening Up America Again


